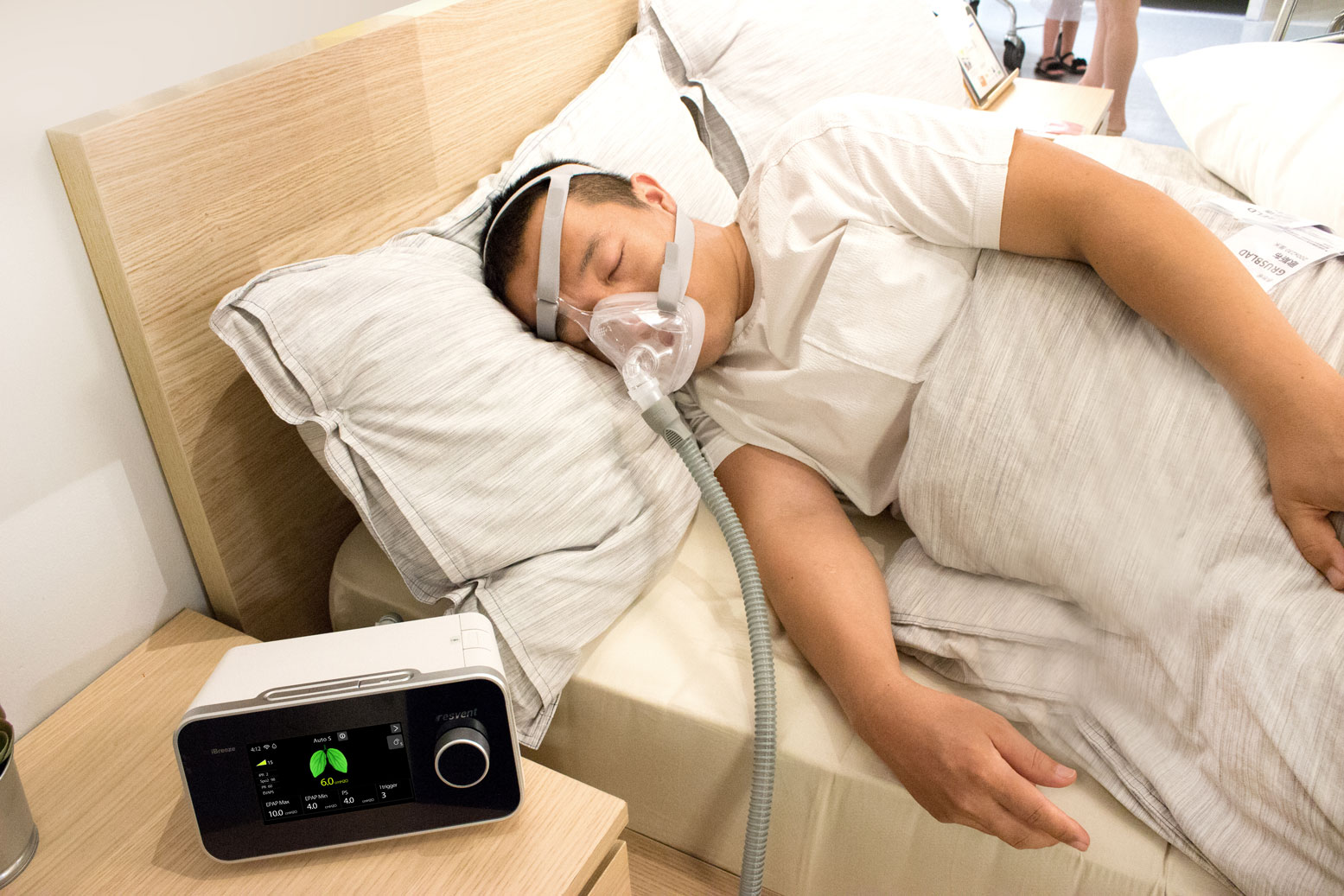
How Connected CPAP and Oxygen Concentrators Transform Respiratory Care
Connected CPAP machines and oxygen concentrators allow institutions to remotely monitor therapies, reduce device losses, and ensure higher treatment compliance. Learn how SysMed USA brings GSM/GPRS-enabled devices to hospitals and gas companies across Latin America.

Key Takeaways
- GSM/GPRS-enabled devices transmit therapy data to the cloud without local Wi-Fi — critical for fragmented connectivity across LATAM markets.
- Connected concentrators with geo-tracking prevent equipment theft — institutions in Mexico, Colombia, and Brazil lose hundreds of units annually to non-return.
- ResAssist™ cloud platform enables remote AHI monitoring, pressure adjustment, and compliance reporting without field visits.
- Predictive maintenance alerts (low O₂ purity, compressor issues) reduce downtime and service costs for distributors managing large device fleets.
- SysMed USA offers GSM-connected oxygen concentrators — among the few available globally — with a zero-recall track record.
In Latin America, where healthcare institutions and industrial gas companies manage thousands of respiratory therapy devices, the challenges go beyond procurement cost. Hospitals and distributors must ensure that patients comply with prescribed therapies, minimize losses from equipment theft, and streamline technical support across wide geographies.
This is where connected CPAP machines and oxygen concentrators, equipped with GSM/GPRS and cloud integration, are reshaping the industry. By enabling real-time monitoring, secure data transfer, and predictive maintenance, these devices allow procurement teams to deliver not only equipment, but also measurable improvements in therapy outcomes.
At SysMed USA, we provide FDA- and CE-certified connected respiratory devices such as the iBreeze PAP series and next-generation oxygen concentrators, purpose-built for institutional buyers across Latin America.
What Does “Connected” Really Mean in CPAP and Oxygen Devices?
Connectivity in respiratory equipment is not just about Wi-Fi, it’s about seamless, secure integration with clinical workflows.
- GSM/GPRS Transmission: Devices automatically send usage and therapy data to the cloud without requiring local Wi-Fi, critical in regions where connectivity is fragmented.
- ResAssist™ Cloud Platform: Resvent’s ecosystem allows clinicians to track key metrics such as AHI (Apnea-Hypopnea Index), CAI (Central Apnea Index), tidal volume, and leak rates in real time.
- Remote Data Access: Reports can be downloaded, exported, or shared directly with insurers, national health programs, or internal compliance teams.
- Modular Connectivity Options: Buyers can choose from Wi-Fi, SIM-based cellular modules, or SD card storage depending on infrastructure needs.
Why Connectivity Matters for Procurement in Latin America
1. Remote Monitoring & Compliance Assurance
Latin American healthcare systems often struggle with follow-up compliance due to geographic dispersion. Connected CPAP and concentrators automatically record adherence data, enabling clinicians to:
- Verify therapy compliance remotely.
- Adjust pressure settings without in-person intervention (via ResAssist remote prescription tools).
- Identify early warning signs of therapy failure.
This reduces patient risk and ensures that institutional contracts tied to compliance metrics are met.
2. Preventing Equipment Loss and Theft
According to SysMed USA client feedback, hundreds of concentrators are lost annually in markets like Mexico, Colombia, and Brazil due to patient non-return or theft.
- GSM-enabled devices provide geo-tracking capabilities, allowing institutions to recover misplaced equipment.
- Alerts can flag when devices are disconnected, tampered with, or inactive for long periods.
This protects multimillion-dollar procurement investments and lowers total cost of ownership.
3. Operational Efficiency & Lower Service Costs
Traditional device management requires costly field visits for audits, recalibration, or data collection. Connected devices:
- Eliminate the need for frequent site visits.
- Allow technical teams in Mexico and Colombia to provide remote onboarding and troubleshooting.
- Reduce downtime with predictive alerts (e.g., low O₂ concentration alarms on SysMed’s M50 concentrator).
The result: fewer interruptions, faster ROI, and scalable operations for regional distributors.
4. Enhanced Patient Outcomes
Features like EVAPS (Essential Volume Assured Pressure Support) ensure tidal volume delivery even in severe cases. Combined with intelligent humidification and IPR (Intelligent Pressure Release), connected CPAPs guarantee:
- More comfortable therapy.
- Fewer dropouts due to dryness or mask discomfort.
- Continuous optimization of therapy pressure in real time.
Better outcomes mean stronger institutional reputations and improved patient retention in long-term oxygen therapy programs.
SysMed USA’s Advantage in Connected Devices
Unlike many global competitors, SysMed USA offers connected solutions tailored to the realities of Latin America:
- Exclusive Distribution of Resvent iBreeze Series (CPAP, APAP, BPAP) with GSM/Wi-Fi connectivity.
- Connected Oxygen Concentrators with GSM modules, among the few available worldwide.
- Zero Recall History, compared to Philips’ high-profile CPAP recalls affecting millions globally.
- Comprehensive Certifications: FDA EUA, CE, ISO 13485, HIPAA, and GDPR compliance.
- Local Technical Presence: Certified technicians in Mexico and Colombia provide onboarding, training, and follow-up support.
- Trusted by Global Leaders: SysMed serves clients such as Linde, Messer, and Air Liquide.
Conclusion
For institutional buyers in Mexico, Colombia, Brazil, Argentina, and across Latin America, connected CPAP and oxygen concentrators are not just innovations, they are necessities. They ensure therapy compliance, asset security, operational efficiency, and better patient outcomes.
By partnering with SysMed USA, procurement teams secure certified, connected, and recall-free solutions, backed by technical support and regional expertise.
References
- Resvent Medical Technology — ResAssist™ Cloud Platform Technical Documentation
- SysMed USA — Client Feedback Data on Equipment Loss Rates in LATAM Markets (2024)
- U.S. Food & Drug Administration — Emergency Use Authorization Framework for Medical Devices
- European Commission — CE Marking Directive for Medical Devices (MDR 2017/745)
- ISO 13485:2016 — Medical Devices Quality Management Systems
Frequently Asked Questions
How does ResAssist™ cloud connectivity work on the iBreeze CPAP/APAP, and what data does it capture?
ResAssist™ is Resvent's proprietary cloud platform for remote patient monitoring and therapy management. The iBreeze transmits nightly therapy data — AHI (apnea-hypopnea index), mask leak rates, usage hours, pressure levels, and tidal volume — via optional WiFi or GSM connectivity to a secure dashboard accessible by clinicians and DME providers.
For distributors, this is a selling point that differentiates against offline-only competitors. Clinicians can monitor patient compliance remotely, adjust pressure settings without an office visit, and generate adherence reports for insurance documentation. The platform supports fleet management across multiple patients, which matters for large DME operations and hospital sleep programs managing hundreds of active therapy cases.
What is the difference between APAP and fixed CPAP modes on the iBreeze, and which do most distributors stock?
Fixed CPAP delivers a single, constant pressure throughout the night — set it once and it stays there. APAP (auto-adjusting) continuously monitors breathing patterns and adjusts pressure breath-by-breath within a clinician-set range, responding to positional changes, REM sleep, and airway events in real time.
Most wholesale partners stock both configurations, but APAP outsells fixed CPAP roughly 3:1 in LATAM markets. The reason is practical: APAP requires fewer titration studies to dial in, which matters in markets where sleep lab access is limited. Clinicians can set a wider pressure range and let the algorithm optimize, reducing return visits and improving patient adherence rates.
What oxygen purity does the O2 Tiger M50 deliver, and at what flow rates?
The M50 delivers 93% ±3% oxygen purity at its maximum flow rate of 5 LPM (liters per minute). Purity remains stable across the full 1–5 LPM range, making it suitable for patients prescribed continuous low-flow oxygen therapy.
For context, 93% ±3% meets the ISO 80601-2-69 standard for oxygen concentrators and falls within the therapeutic range for most long-term oxygen therapy (LTOT) prescriptions. The concentrator uses molecular sieve technology (pressure swing adsorption) to separate oxygen from ambient air — no tanks, no refills, no delivery logistics. For distributors serving homecare providers and hospital discharge programs, the M50 covers the majority of LTOT patients who need 1–5 LPM continuous flow.
What does FDA 510(k) clearance mean for the respiratory devices SysMed distributes?
FDA 510(k) clearance means the U.S. Food and Drug Administration has reviewed the device and determined it is substantially equivalent to a legally marketed predicate device in safety, intended use, and performance. Every Resvent device SysMed distributes — ventilators, CPAP, auto-CPAP, and BiPAP systems — holds active 510(k) clearance.
For international buyers, this matters because several LATAM regulatory agencies (COFEPRIS, ISP, MINSA) accept FDA clearance as supporting evidence in their own registration processes, which can shorten approval timelines significantly. It also signals that the device meets manufacturing quality standards under FDA's Quality System Regulation (21 CFR 820), backed by ISO 13485 certification.